- Temperature - related to average KE of molecules
- Fahrenheit scale
- Celsius scale
- Kelvin scale
- Internal Energy - total KE and PE of molecules
- Heat - transfer of internal energy due to a temperature difference
- compare to Work - transfer of energy due to a force
- If there is thermal contact, heat "flows" from high temperature to low temperature until thermal equilibrium is reached.
- 1 calorie = heat to raise temperature of 1 gram of water by 1o Celsius = 4.2 Joules
- 1 Calorie = 1000 cal (diet calorie)
- Specific Heat Capacity
- amount of energy need to raise the temperature of 1 gram of a substance by 1oC
- high specific heat capacity
- lots of energy required to raise the temperature
- lots of energy available at high temperature
- Water's specific heat capacity is high (1 cal/gramoC)
- low specific heat capacity
- large rise in temperature for a little added energy
- not much heat energy available even at high temperatures
- Solving heat transfer problems
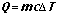
- Example
- Expansion
- most substances expand when heated
- solids least, gasses most
- water's behavior is unusual
- maximum density at 4oC
- pond freezing
- maximum density at 4oC
- most substances expand when heated
- Demo: Expansion of Gasses, Liquids, and Solids
- CPSurf.com - annotated links keyed to your textbook